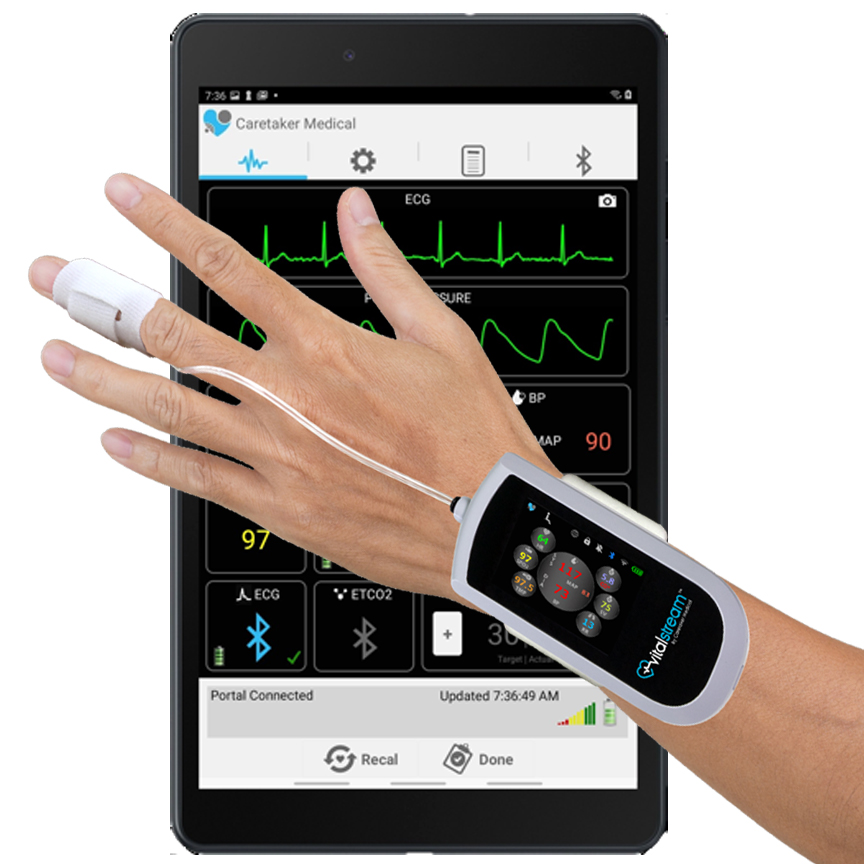
Setup
NIBP Unit remains in control room and tubing passes through the waveguide to subject.
System Components
- MRI-Compatible “VitalStream” Continuous Monitor
- Finger Cuff Transducers x 2
- In general, Cuffs last ~100 hours of intermittent MRI use (max 3-5 days of continuous use)
- additional or replacement sensors available as RXNIBP-A-SENSOR
- Tubing 8 m (25′)
- Tablet computer
- Automatic Blood Pressure Calibration Unit
- Export Utility
- Bluetooth dongle
- Power Supply
- 1-Year Warranty
Calibration
The VitalStream device can be calibrated automatically using its self-calibration mode or manually with an included cuff. An export conversion utility is included to import calibrated pulse wave data to AcqKnowledge research software.
Part #: NIBP-B-MRI





Stay Connected