H01 12-Lead ECG
This BSL PRO lesson describes the hardware and software setup necessary to record a 12-lead ECG.
Experimental Objectives
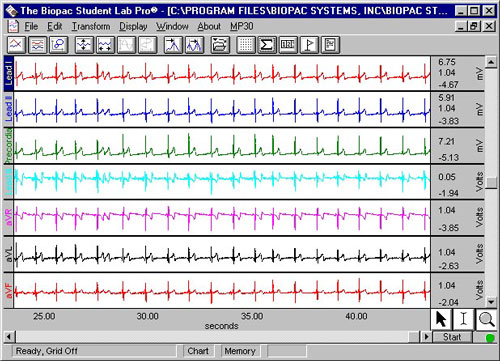
- To record each lead of the standard 12- lead ECG (Lead I, Lead II, Lead III, aVR, aVL, aVF, V1, V2, V3, V4, V5, V6).
- To observe changes in the frontal plane vectors throughout a cardiac cycle.
Tasks Performed by the Student
You will record six 10-second segments, for a total recording of 60 seconds. You will record Lead I, Lead II and a Precordial (chest) lead in six positions (V1 through V6). In addition, Lead III and the “Augmented leads” (aVr, aVl, aVf) will be calculated. Each chest electrode is positive and the whole body is used as the common ground.
Videos
Biopac Student Lab Student Download
Student Prep & Distance Learning
Click the link(s) below for sample data and/or lesson procedure video(s), BSL PRO Lesson procedures (PDF) for human lessons*, and graph template files (*.gtl) for BSL PRO Lessons. If more than one .gtl is available, download the .gtl with the _suffix to match BSL version and hardware.
Lesson Hardware
This lesson requires a Biopac Student Lab (BSL) System and the following hardware. If your BSL System does not include all hardware items, expand your system by selecting required items below. For more details, review the Lesson: L# BSL Lessons - see the Lab Manual or launch BSL; A# and H# BSL PRO Lessons, click the PDF link above to review full setup, recording, and analysis procedures.
| Item | Name | Cart |
|---|---|---|
| SS29L | Multi-lead ECG Cable (MP3X) | Add to Cart |
| EL503 | Disp. Gen-purp electrode 100/pk | Add to Cart |
| ELPAD | Abrasive Pads 10/pk | Add to Cart |
| GEL1 | Electrode gel 50 g | Add to Cart |
Stay Connected